Chiral molecules reveal their handedness
An international group of scientists around Melanie Schnell from CFEL and Harvard performed the first chirality-sensitive broadband microwave spectroscopic analysis of a chiral mixture.
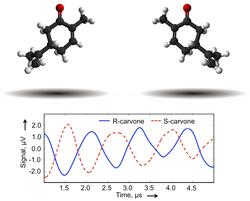
Left- and right-handed enantiomers of carvone (top) and their differentiation via different phases as a result of microwave three-wave mixing (bottom).
The chemistry of life is built from left-handed and right-handed molecules that are mirror images of each other. Such molecules are said to be chiral, a word derived from the ancient Greek word for hand, cheiros. The two molecules in a chiral pair are called enantiomers and can have completely different biological properties. For example, the right-handed enantiomer of carvone smells like spearmint while the left-handed enantiomer smells like caraway. Chiral molecules often occur in complex mixtures such as medicines and perfumes. Analyzing these mixtures to identify the molecular components, determine their handedness, and measure their relative amounts (i.e., the enantiomeric excess) is still one of the most challenging and very important tasks in analytical chemistry. These analyses are needed in essentially all phases of modern drug development, from early candidate searches to production and regulation.
An international team of scientists from the Max Planck Institute for the Structure and Dynamics of Matter at the Center for Free-Electron Laser Science Hamburg, the Hamburg Centre for Ultrafast Imaging, and the Department of Physics at Harvard University in Cambridge, have performed the first chirality-sensitive broadband microwave spectroscopic analysis of a chiral mixture. They achieved this analysis using a conformational mixture of carvone and report it in the Angewande Chemie. An experimental highlight is the determination of the absolute configurations (i.e. handedness) of the enantiomers. The method employs microwave three-wave mixing and is inherently mixture-compatible, a unique advantage over other techniques. This advantage arises from the fingerprint-like character of the rotational spectra of molecules. In future experiments, the scientists will extend the technique towards analyzing larger and more complex molecules and samples of biological relevance.