X-ray pulses reveal structure of viral cocoon
An international team of scientists has used high-intensity X-ray pulses to determine the structure of the crystalline protein envelope of an insect virus. Their analysis reveals the fine details of the building blocks that make up the viral cocoon down to a scale of 0.2 nanometers (millionths of a millimeter) – approaching atom-scale resolution. The tiny viruses with their crystal casing are by far the smallest protein crystals ever analyzed using X-ray crystallography. This opens up new opportunities in the study of protein structures, as the team headed by DESY’s Leading Scientist Henry Chapman from CFEL reports in the current issue of Proceedings of the U.S. National Academy of Sciences (PNAS).
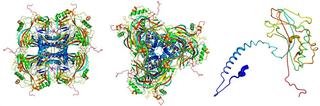
Atomic model of the crystalline occlusion bodies, derived from the X-ray diffraction images recorded at the LCLS. The individual proteins (right) stick together to form the building blocks (left, seen from the side; center, seen from above) of the crystalline occlusion bodies. Credit: Dominik Oberthür, CFEL/DESY
“The granulovirus attacks certain insects and kills them. This initially leaves it stranded inside the decaying host, so it has to protect itself, perhaps for years, against adverse environmental conditions such as heat, ultraviolet radiation and drought, until it is once again ingested by an insect. To achieve this, the virus wraps itself in a cocoon made of protein crystals, which only dissolve again once it reaches an insect’s gut,” explains Cornelius Gati from DESY, the main author of the paper. These viruses are a particular interest of Peter Metcalf from the University of Auckland in New Zealand and Johannes Jehle from the Julius Kühn Institute in Darmstadt, who teamed up with DESY for this research. The researchers examined the cocoon of the Cydia pomonella granulovirus (CpGV), which infects the caterpillars of the codling moth (Cydia pomonella) and is used in agriculture as a biological pesticide. The virus is harmless to humans.
Scientists are interested in the spatial structure of proteins and other biomolecules because this sheds light on the precise way in which they work. This has led to a specialized science known as structural biology. “Over the past 50 years, scientists have determined the structures of more than 100,000 proteins,” says Chapman, who is also a professor of physics at the University of Hamburg. “By far the most important tool for this is X-ray crystallography.” In this method, a crystal of the protein under investigation is grown and irradiated with bright X-rays. This produces a characteristic diffraction pattern, from which the spatial structure of the crystal and its building blocks can be calculated.
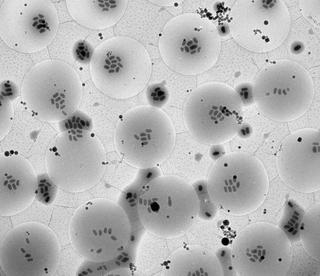
Transmission electron microscope image of granulovirus particles on an object grid. The field of view measures approximately 50 microns (0,05 millimeters). Credit: Ken Goldie, C-CINA Basel
Scanning electron microscope image showing the homogeneous size of the virus particles. Field of View is about 5 microns. Credit: Peter Metcalf, University of Auckland
“One of the big challenges of this procedure is, however, growing the crystals,” adds Chapman. Many proteins do not readily align to form crystals, because that is not their natural state. The smaller the crystals that can be used for the analysis, the easier it is to grow them, but the harder it is to measure them. “We are hoping that in future we will be able to dispense altogether with growing crystals and study individual molecules directly using X-rays,” says Chapman, “so we would like to understand the limits”.
“These virus particles provided us with the smallest protein crystals ever used for X-ray structure analysis,” explains Gati. The occlusion body (the virus “cocoon”) has a volume of around 0.01 cubic micrometers, about one hundred times smaller than the smallest artificially grown protein crystals that have until now been analyzed using crystallographic techniques.
To break this limit in crystal size, an extremely bright X-ray beam was needed, which was obtained using a so-called free-electron laser (FEL), in which a beam of high-speed electrons is guided through a magnetic undulator causing them to emit laser-like X-ray pulses.
The scientists used the free-electron laser LCLS at the SLAC National Accelerator Laboratory in the U.S., and employed optics to focus each X-ray pulse to a similar size as one of the virus particles. “Directing the entire power of the FEL onto one tiny virus exposed it to the tremendous radiation levels,” reports Gati, who now works at SLAC. The dose was 1.3 billion Grays; for comparison: the lethal dose for humans is around 50 Grays.
The FEL dose was certainly lethal for the viruses too – each was completely vaporized by a single X-ray pulse. But the femtosecond-duration pulse carries the information of the pristine structure to the detector and the destruction of the virus occurs only after the passage of the pulse. The analysis of the recorded diffraction showed that even tiny protein crystals which are bombarded with extremely high radiation doses can still reveal their structure on an atomic scale.
“Simulations based on our measurements suggest that our method can probably be used to determine the structure of even smaller crystals consisting of only hundreds or thousands of molecules,” reports Chapman, who is also a member of the Hamburg Center for Ultrafast Imaging (CUI). “This takes us a huge step further towards our goal of analyzing individual molecules.”
The European XFEL GmbH in Schenefeld, the Max Planck Institute for Medical Research in Heidelberg, the Arizona State University (USA) and the University of Basel (Switzerland) were also involved in the research.